Application note - Centralized biobank structures in
university hospitals

High-quality biospecimens and associated
medical information for modern biomedical research
Biospecimens hold significant value for modern biomedical research. There is consensus within the medical-scientific community that biobanks can significantly contribute to uncovering the causes, mechanisms, and influencing factors of diseases, enabling more precise and reliable diagnoses and more effective treatments. The quality of human biospecimens and their associated data is
of utmost importance for achieving reliable and reproducible scientific results. To ensure this, all subprocesses within biobanks, such as acquisition, processing, storage, and distribution of biospecimens, must be executed with the highest quality standards and transparently documented.
The LVL SAFE® 2D Tubes enable verifiably secure and space-saving storage of various types of samples, as well as secure identifiability. In addition to being suitable for manual laboratory processes, the products are also excellent for automated processing and storage across all temperature ranges. They exhibit high compatibility with existing systems. An optimal workflow
is achieved through the use of LVL SAFE® 2D Barcode Scanners (for automated code scanning of tubes and racks) and LVL SAFE® 8-Channel and 96-Channel Cappers/Decappers (for opening and closing the tubes). These products are used in biobanks worldwide.
Zebanc
– Central biobank Charité –
in the largest university hospital in Europe
The Central Biobank Charité – abbreviated as ZeBanC – is a joint core facility of the Charité and the Berlin Institute of Health at Charité (BIH). It is a clinically oriented biobank for the quality-controlled storage of biospecimens derived from clinical processes. In close collaboration with clinical colleagues, ZeBanC assists in the processing and storage of biospecimens (tissues, fluids, derivatives) and provides samples for translational research projects. The biobank’s quality management system is certified according to DIN EN ISO 9001:2015.
The use of biospecimens has already contributed to new research insights in numerous studies. Selected project examples include the National Pandemic Cohort Network (NAPKON), a high-quality cohort collection of patients with the coronavirus disease; TransBioline, a European study on biomarker research; and the BeLOVE study (Berlin Long-term Observation of Vascular Events) of the BIH, a cohort study investigating cardiovascular diseases.
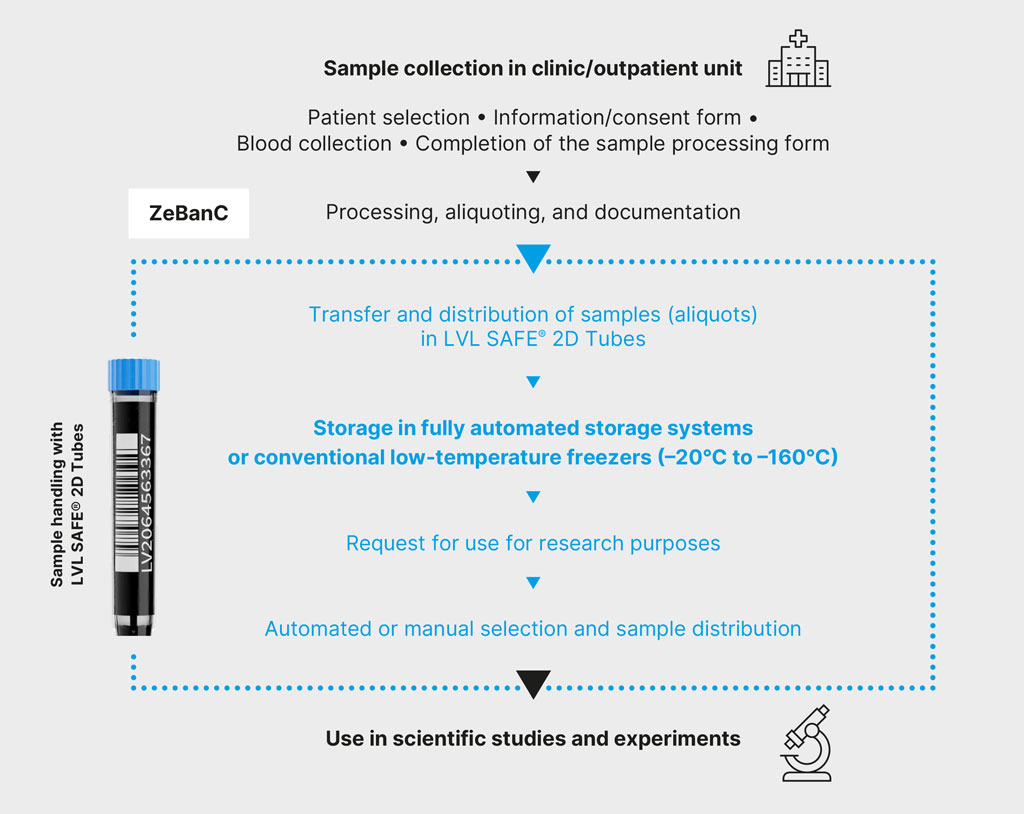

Samples & Processes
- Sample collection in the clinic after obtaining patient consent.
- Transfer of samples (e.g., EDTA blood, serum) along with a completed sample processing form to ZeBanC.
- Processing (e.g., centrifugation, PBMC isolation) of the sample and aliquoting into LVL SAFE® 2D Tubes, for which pre-prepared, study-specific aliquoting sets with tubes and racks are provided by ZeBanC staff; use of structured color schemes for the tube caps for clearly identifiable sample structures to simplify processes and reduce error susceptibility.
- Storage in various storage systems:
- automated storage system at –80°C OR
- automated storage system at –160°C in the gas phase of liquid nitrogen OR
- conventional freezers or tanks (–20°C to –160°C)
- Documentation of the sample (unique sample ID using barcode), preanalytical parameters, and other associated data in a biobank information system.
- All relevant information is linked to the code on the tubes in the database: patient assignment, sample type, sample quantity, collection timestamp, processing, storage time, storage location, etc.
Customer feedback on the use of products in biobanks –
User experience & benefits
Well-thought-out product design offers a variety of user benefits
- Pseudonymization of samples, unique sample ID, and clear labeling – no self-labeled samples with patient names,
no risk of adhesive labels falling off. - Efficiency improvement through scanning of 2D codes during sample processing compared to traditional reaction
vessels, where manual assignment and verification were/are necessary. - Tubes are available in various sizes and with different cap colors, making them well-suited for different studies and
requirements – th range remains clear and manageable. - Significant space savings compared to sample systems with traditional reaction vessels in, for example, cardboard boxes.
- Time savings through use of the capper/decapper when opening and closing an entire rack – all tubes in a rack or row are
automatically opened or closed in one operation.
Compatibility with equipment throughout the entire process
- Availability of technical drawings for the tubes and racks with precise dimensions is very helpful for evaluating suitability
for use and estimating compatibility with, for example, automated systems. - Standard SBS formats of the tubes lead to high compatibility with other equipment used in the process chain; direct pipetting/transfer into 96-well microtiter plates is possible.
- Automation-friendliness – good compatibility with automated systems in the process (e.g., pipetting robots, automated
storage systems).
Reliable product and delivery quality as a prerequisite for long-term use
- Accredited data on product quality provided in advance: certificates from accredited institutes on the seal integrity
(IATA/CO2) and on the leachables and extractables of the tubes. - Stable product quality of the vessels, both for immediate use and for long-term storage and subsequent use in
future studies – long-term supply capability ensured. - Reuse of racks, purchase of bulk goods for sustainability.
- Fast and reliable delivery.
- Professional, pleasant communication, quick response time from LVL staff, short communication channels.
Referenzen & Kontakt

Dr. Jenny Schlesinger
Central Biobank Charité (ZeBanC)
Scientific coordination

Dr. Myriam Merz
Central Biobank Charité (ZeBanC)
Scientific coordination
Webseite
https://biobank.charite.de/en




