Application note - Modern archiving of blood
donation plasma samples in
blood banks
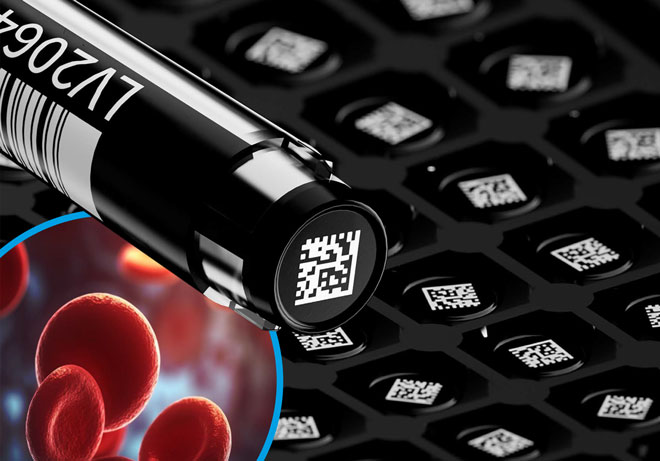
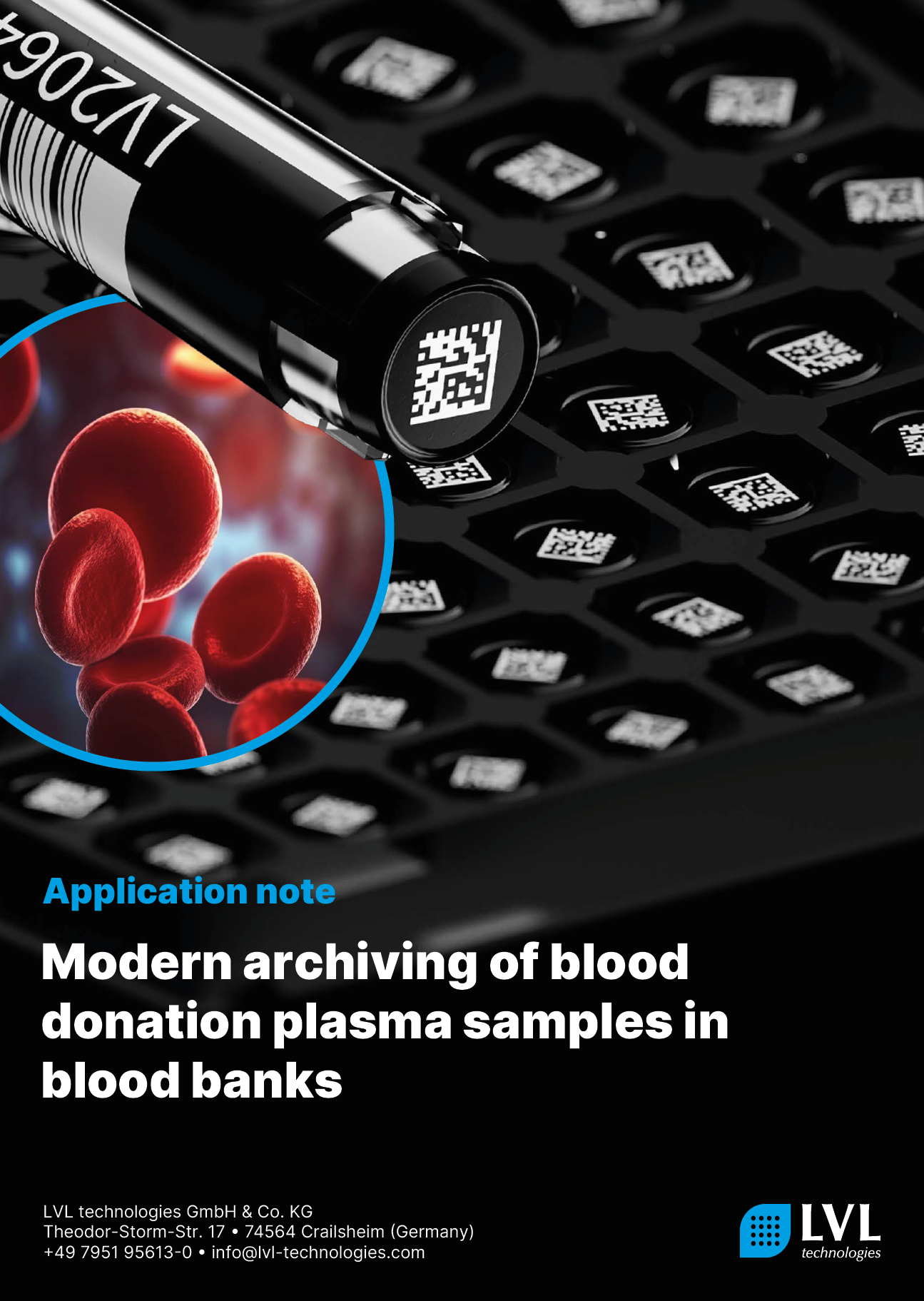
Archiving samples in blood banks is essential
The processing and deep frozen storage of archived samples for each blood donation is compulsory for blood transfusion services in Germany. The samples must be stored for an additional year beyond the shelf life of the blood products obtained from the blood donations. These archived samples must be suitable for all analytical tests applied in the context of look-back procedures. Additionally, the National Advisory Committee “Blood” (Arbeitskreis Blut), an advisory board for German medical authorities, recommends in its vote 47 that blood donation facilities should archive at least 1.5 ml of reserve volume at < –30°C. The samples should be stored in a manner that ensures they can be further examined using Nucleic Acid Amplification Techniques (NAT). Great emphasis is placed on the unequivocal identification of each sample based on its individual container.
These requirements can be met by means of new processes utilizing LVL products. The following is an exemplary description of the process by the Central Laboratory of the Blood Donation Service West in Hagen.
A practical report
DRK Blood Donation Service West – Central Laboratory Hagen
The transition took place in the Central Laboratory of the Blood Donation Service West in 2022. The entire process of creating, managing, and processing archive samples from each blood donation (approximately 750,000 per year) was aligned with the national guidelines of the National Advisory Committee “Blood” (Arbeitskreis Blut). The long-term security of samples has been improved and the process is now considerably less time-consuming, both during storage and in the case of withdrawals.
Process step - Removal and storage
- Centrifugation and automatic uncapping of a 10-ml EDTA plasma tube
- Preparation of 2 aliquots of 1 ml EDTA plasma in LVL SAFE® 96 IT: 2D tubes with internal thread in 96 SBS (Society for Biomolecular Sciences) format while monitoring correct aspiration and liquid delivery in a liquid handling platform/pooling system
- Sealing the 2D tubes with a SAFE® Cap Sealer with SAFE® Push Cap Mats
- Linking 2D codes of processed tubes on the rack with the barcode of the tube rack and the storage tray using the userfriendly
tube rack reader LVL SAFE® READ Single Express - Export of data via an interface to an internal company database
- Transport of the loaded storage trays to a freezing cell (–40°C)
- Storage of EDTA plasma aliquots in mobile stacking racks for 5 years
Process step - Use for traceability procedures
- Database query by entering the donation ID, i.e. coordinates for removing the archived sample aliquot
- Rapid on-site identification of the sample using the SAFE® READ
Single Code HD in combination with the Tube Presenter 96 - Easy and quick retrieval of individual samples from the storage system
- Use in analytics without merging individual aliquots
To ensure continuous look-back analytics, parallel storage in the predecessor system is carried out for the necessary transition period of 4 years.
The specifications from the National Advisory Committee „Blood“ in vote 47 (05/2019) and vote 48 (11/2020) have been implemented
Look-back samples can be analyzed using individual donation NAT (Nucleic Acid Amplification Techniques).
Individual tubes are uniquely identifiable. The transfer and storage of look-back samples are done in individual tubes with 2D coding, featuring a double ID. The tubes are pre-coded with a batch ID and additionally, each individual sample has its unique and unmistakable identification. This method ensures clear and unambiguous identification, even in the case of simultaneous withdrawal of multiple samples or accidental operational errors (e. g., a rack falling down).
BEFORE: 1 : 1 labeling of the sample only via barcode of the entire microtiter plate (MTP) and assignment via data management.
The volume required for each look-back sample per donation has been increased to a minimum of 1.5 ml, divided into 2 aliquots for each blood component donation.
Separation of 2 x 1 ml per donation.
BEFORE: Separation of 5 x 0.2 ml per donation.
Merging of the individual aliquots is not necessary for reprocessing.
Retesting is ensured with the available volume of 1 ml; for each PCR analysis, a minimum of 0.8 ml is required.
The retrieval and removal of both the racks and the individual samples is significantly faster due to easy access to the racks in the storage systems. The lid cover of the LVL racks prevents the tubes from being rubbed – making them much easier to read.
This significantly reduces the overall time required for the process, not least for the staff in the deep-freeze warehouse (health benefits).
BEFORE: Tedious retrieval of the appropriate microtiter plate from a fully stacked collapsible plastic box, followed by timeconsuming emptying and restacking of all other microtiter plates into the box.
No manual coding of the racks – the racks from LVL are already pre-coded.
BEFORE: Each individual rack had to be labeled manually with a paper barcode.
Further aspects of increased safety and sample quality
Greater certainty regarding sample identification and allocation.
Identification is more reliable due to the good legibility. The irrelevant samples remain unopened.
BEFORE: Sealing was carried out manually using an adhesive film. If an analysis was required, the cover had to be removed from all 96 wells of the MTP.
Minimized risk of loss of target nucleic acid for NAT due to degradation effects and the associated false-negative NAT
result.
Individual tubes from the SBS rack can be thawed again under standardized conditions, which reduces the risk of degradation
of the pathogen‘s nucleic acids.
BEFORE: Simultaneous and unnecessary thawing and refreezing of all samples in the 96-well microtiter plate rack were
necessary.
High level of security regarding the long-term maintenance of product quality due to the durability of racks and sample tubes.
LVL ensures to an exceptionally high degree that no material defects and associated sample losses can occur during processing and years of storage:
- Each tube undergoes a leak test
- Certificates for compatibility with automation systems are available
- The racks‘ dimensional accuracy is maintained
- Fatigue-resistant materials are utilized
- Database comparison is conducted to prevent duplicates in terms of coding
Enhanced security in the event of a freezer cell failure.
In the event of a technical malfunction of the freezer cell/area archive, samples can be easily transferred to a backup device, thereby preventing inadvertent thawing of the samples.
Lower capacities of expensive freezer volume are required.
The racks are stored in a very compact form.
Automated process steps are possible – including expansion to a fully automated storage system.
The racks from LVL comply with standards regarding dimensions (SBS) and coding procedures (ISO 15415), making them compatible with all automation systems.
Conclusion
The newly introduced process of storing and managing archived blood bank samples in appropriate containers, as previously described, aligns with current national guidelines, is efficient, easy to handle, significantly reduces workload, and opens possibilities
for further automation.
The collaboration with LVL has been excellent. We are very satisfied and look forward to the potential expansion towards a fully automated storage system.
Customer feedback
User experience & benefits
Communication, support, and delivery reliability from LVL ensure the optimal process.
The company exhibits professional expertise with a deep understanding of processes. We have valued the direct, fact-based, and solution-oriented communication with the company for many years, along with the continuous optimization of its products and processes.
The support is highly reliable. In case of any emerging questions or issues, there is a prompt response and excellent service. Delivery reliability, as an essential prerequisite for the long-term utilization of such a system, is guaranteed.