Biobanking for the Population-based Study on
Health and Living Conditions in Regions with
Sámi and Norwegian Population

SAMINOR 3 – UiT The Arctic University of Norway
The Sámi people are the indigenous population of the North. In 2003, the collection of biological samples and information on the health and lifestyle of the Sámi community commenced in Norway in the population-based SAMINOR Study. The aims of the study are to gain insight into the preva-lence and risk factors for diseases, to prevent illness, and to improve healthcare for all.
At the 50 regionally distributed sampling stations now conducting SAMINOR 3, which started in 2023, the samples are being collected directly into LVL SAFE® 2D Tubes. The high suitability of the tubes for both manual handling at the sampling stations and automated processing and storage of the samples was a crucial selection criterion. The pre-labeling of the tubes and racks, as well as the automation of the scanning, reading, capping, and decapping, led to significant time savings in the SAMINOR 3 study compared to the entirely manual handling and labeling during SAMINOR 1 and 2. The risk of human error in handling is significantly reduced with the new products and processes. Automated storage systems will be introduced in the next step.
Unique Database – One of the most important sources
of knowledge on the health and living conditions in the
Sámi and Norwegian populations of rural northern Norway
This SAMINOR Study – The Population-based Study on Health and Living Conditions in Regions with Sámi and Norwegian Populations – places particular emphasis on the health and living conditions of the Sámi community, rendering SAMINOR a distinctive database both nationally and internationally due to its Indigenous perspective. The Centre for Sámi Health Research (CSHR) oversees the SAMINOR Study and collaborates with various research teams engaged in health research within Indigenous communities across Canada, the USA, Australia, New Zealand, Denmark/Greenland, and Sweden. Blood samples are stored in the Core Facility for Biobank at UiT The Arctic University of Norway.
The SAMINOR Study is a collection of surveys conducted approximately every 10 years. The surveys to date include: the SAMINOR 1 Survey (2003–2004), the SAMINOR 2 questionnaire Survey (2012), and the SAMINOR 2 Clinical Survey (2012–2014). Between 2003 and 2014, SAMINOR 1 and 2 collected data from over 30,000 individuals, whereof approximately 18,000 have donated blood samples to the biobank one or two times. Now, between 2023 and 2025, SAMINOR 3 will integrate an additional 72,000
participants from across Norway aged between 40 and 79. The SAMINOR 3 Survey, which consists of an interview survey, is already completed. The clinical ide is currently in progress. At the same time, within the framework of SAMINOR 3, the biobank is taking its first steps toward automation.
All researchers (PhD level or equivalent) at Norwegian research institutions can apply to analyze data from SAMINOR. The use of blood samples requires approval by REC North (Regional Committee for Medical and Health Research Ethics), collective Sámi consent from the Expert Ethics Committee for Sámi Health Research, and by the SAMINOR project board, which advises the SAMINOR director.
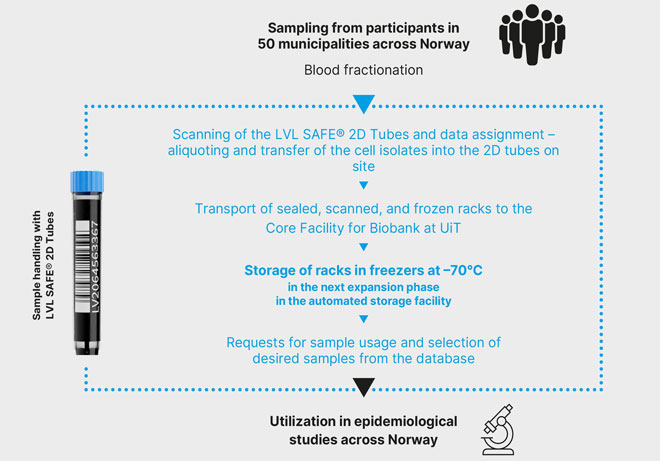

- Participants are successively invited to a clinical examination on the bus or to the temporary research station, which gathers information on living conditions, lifestyle, health, etc., using a standardized questionnaire, medical examinations, and collection of blood, urine, and hair samples.
- Standardization of sampling conditions is achieved by deploying the same personnel, who travel to the respective stations. At any time, there are two mobile research units collecting samples at different locations, usually in two different municipalities. One research unit moved from place to place on a trailer, whereas the other is a research bus (named “Odin”).
- The processing of the samples includes cell isolation using centrifugation, which fractionates whole blood into its component parts, such as buffy coat and plasma.
- Eight samples per participant (3 plasma, 4 serum, 1 leukocyte) are transferred into the 1000 µl LVL SAFE® 2D Tubes, one row per SBS rack per participant, 12 participants in one rack.
- A fixed color concept is utilized for caps to ensure higher allocation security; LVL customizes delivery accordingly.
- The 8-Channel Capper/Decapper is used for capping and decapping in all process steps. Tubes and racks are scanned.
- The samples are kept in the freezer until shipment of the sealed racks to the biobank.
- On arrival at the biobank, the tubes are re-organized by the color of their lids. The racks are scanned and read again, and the samples are placed in freezers for long-term storage at –70 °C.
- The introduction and evaluation of robotics and automated storage systems to increase the level of process automation is currently underway.
- Via the database, researchers may select and apply for samples from the biobank. The samples are provided in racks.
- All relevant information is assigned to the LVL SAFE® 2D code. At the research stations, each participant is linked with eight 2D codes corresponding to the eight tubes of samples associated with them. Later, this information is collected in a database and further linked to include information from questionnaires and clinical examination.

Product training at UiT. Showing the use of an LVL SAFE® Capper/Decapper to cap 8 tubes with color-customized caps, and an LVL rack reader that identifies each tube and position in the 96-tube rack by reading the 2D code at the bottom of the tubes.

Behind, from left: Kristin Sørensen, Erik Axel Vollan, and Karina Standahl Olsen, who all work at UiT The Arctic University of Norway, and Reza Nour from Nerliens Meszansky. Front, from left: Marita Melhus, Susanna Ragnhild Siri, and Runa Borgund Barnung, who all work at UiT The Arctic University of Norway, and Claudia Emmanuel from Nerliens Meszansky.
Customer Feedback
User experience & benefits
Flexibility of use – specifically the suitability of the products for BOTH manual handling
AND automated processes – was the key selection criterion
- Easy and convenient handling at the sampling stations and for the current manual storage in freezers.
- Good compatibility with the planned expansion to include robotics and storage automation systems.
Flexibility of use – specifically the suitability of the products for BOTH manual handling
AND automated processes – was the key selection criterion
- The 2D pre-labeling and the automatic scanning of tubes and racks result in high security and time savings in sample
handling – benefits for accurate protocols, precise work, low error rates throughout the entire logistics chain. - The option of adding a third code on the tubes allows for expansion – two options could be of interest: delivery from
LVL using a third human-readable code OR in-house coding with an LVL SAFE® Tube Laser Marker. - Customized delivery based on the color distribution of caps in the racks yields high benefits – time savings and risk
reduction for mix-ups/false fillings; yellow caps are used for serum, pink for plasma and blue for buffycoat. - Easy closure of the lids – possible fit in both directions.
- High security of the closures – samples arrive safely and undamaged at the biobank after transport from the stations.
- High time savings due to the effective capping/decapping by the LVL SAFE® 8-Channel Capper/Decapper.
High-quality support services
- Technical expertise of LVL staff.
- Quick and uncomplicated assistance with initially encountered specific issues with the LVL SAFE® Tube and Rack Reader – close support until final resolution and satisfactory outcome.
Deciding on the appropriate volume is a challenge
- During the evaluation phase, the decision was made for 1000 µl tubes as a compromise between available sample
volumes for studies, space, and cost requirements. - There remains uncertainty about whether the volumes will be sufficient for use in the coming years or whether practical application over time will show that the use of smaller or larger tubes, or the storage of the same sample volumes in smaller tubes in higher numbers, would have been more advantageous.
References & Contact

Susanna Ragnhild
Andersdatter Siri
MPH, PhD, Researcher at the
Centre for Sámi Health Research
and Co-Investigator in SAMINOR 3

Runa Borgund
Barnung
Head Engineer at the Core Facility for
Biobank Faculty of Health Science, UiT

Karina Standahl
Olsen
Associate Professor,
Head of the Core Facility for Biobank,
Faculty of Health Science, UiT



